4 results
Three-day antibiotic duration in patients with pneumonia: A sixty-eight–hospital cohort
- Valerie Vaughn, Lindsay Petty, David Ratz, Elizabeth McLaughlin, Tawny Czilok, Jennifer Horowitz, Anurag Malani, Danielle Osterholzer, Scott Flanders, Tejal Gandhi
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s22
-
- Article
-
- You have access Access
- Open access
- Export citation
-
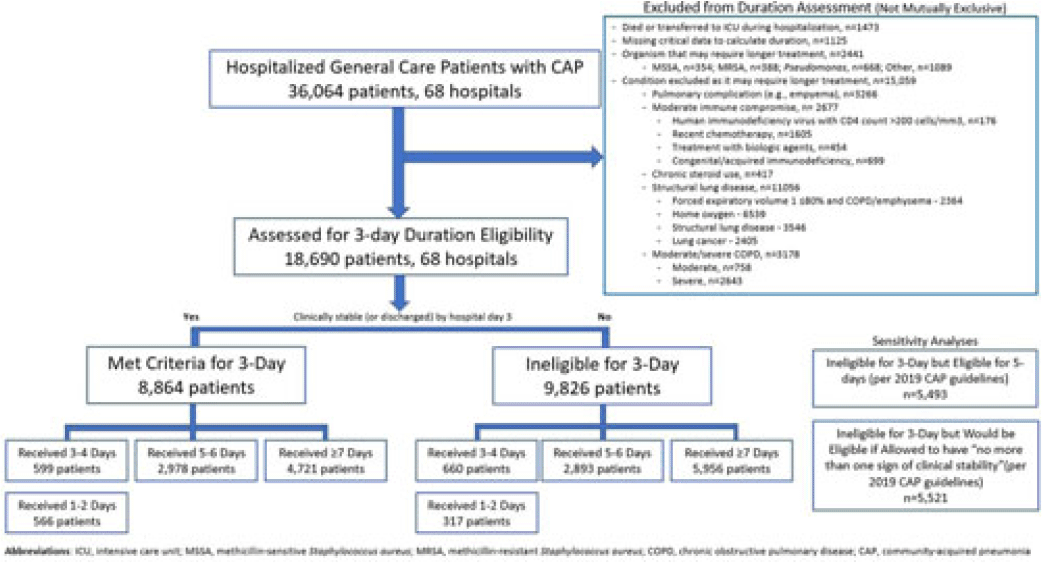
Background: Since 2019, community-acquired pneumonia (CAP) guidelines have recommended hospitalized patients be treated until clinical “stability and for no less than 5 days.” However, randomized trials have reported that, in patients who stabilize by hospital day 3, very short antibiotic durations (eg, 3 days) are noninferior to longer durations. How these trial results relate to real-world practice is unknown. Methods: Using a 68-hospital cohort study of hospitalized, general-care adults with CAP, we aimed to (1) quantify the percentage of patients who—according to trial criteria—qualify for a 3-day antibiotic duration, (2) quantify the percentage who actually received a 3-day duration, and (3) assess 30-day outcomes. Patients were considered to have CAP if they had a pneumonia discharge diagnosis and met clinical criteria for CAP. Patients with concomitant infections (including COVID-19), admission to intensive care, or severe immunocompromise were not included. Results: Between February 23, 2017, and August 3, 2022, 36,064 patients with CAP were included. Of those, 48.2% (9,826 of 36,064) were excluded due to a condition or organism ineligible for the 3-day treatment (Fig. 1). Of the 18,690 patients remaining, 52.6% (9,826) were unstable on day 3 and thus were ineligible for the 3-day treatment. Therefore, of all 36,064 patients, only 8,864 (24.6%) would be eligible under trial criteria for a 3-day treatment. Notably, 5,493 (55.9%) of 9,826 patients unstable on day 3 would be eligible for 5 days of treatment under national guidelines. In practice, use of 3–4-day treatment was rare, occurring in 599 (6.8%) of 8,864 patients eligible for a 3-day treatment versus 660 (6.7%) of 9,826 patients unstable on day 3 (P = .945). Use of 3–4-day treatment increased over time and comorbidities that could mimic CAP or a negative procalcitonin were more common in patients who received a 3–4-day treatment whereas specific symptoms of CAP were less common (Fig. 2). After adjustments, patients eligible for a 3-day duration who received a 3–4 day treatment versus a ≥5-day treatment had higher 30-day mortality (aOR, 1.87; 95% CI, 1.32–2.64) and readmission (aOR, 1.35; 95% CI, 1.17–1.56). Conclusions: Across 68 hospitals, <25% of patients hospitalized with CAP would be eligible for a 3-day antibiotic treatment. Though increasing over time, there was little use of 3–4-day treatments and, when prescribed, outcomes were worse, potentially due to CAP misdiagnosis. Given the small number of patients eligible for 3-day treatment, and the potential harm with too-short durations, it may be prudent to focus on increasing the use of 5-day treatments.
Disclosures: None
Prevalence of and risk factors for bacteremic UTIs in hospitalized adults without definitive signs or symptoms of UTI
- Sonali Advani, David Ratz, Jennifer Horowitz, Lindsay Petty, Kenneth Schmader, Tawny Czilok, Anurag Malani, Tejal Gandhi, Scott Flanders, Valerie Vaughn
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s27-s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
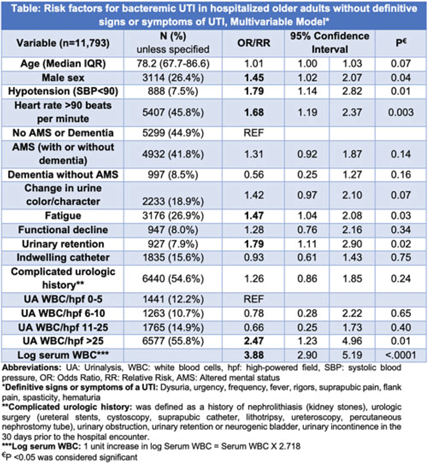
Background: IDSA guidelines recommend withholding treatment in patients with asymptomatic bacteriuria in the absence of systemic signs of infection. However, some patients with bacteriuria may not be able to express symptoms either due to presence of indwelling catheter, underlying complicated urologic anatomy, dementia, or altered mental status (AMS). Clinicians frequently treat bacteriuria in this population with antimicrobial therapy due to concern for sepsis. To determine treatment need, we aimed to review prevalence and risk factors for bacteremic urinary tract infection (UTI) in a cohort of hospitalized inpatients without definitive signs and symptoms of a UTI. Methods: This retrospective cohort study of inpatients with a positive urine culture who presented without definitive signs or symptoms of a UTI was conducted between July 1, 2017, and June 30, 2022, in 68 academic and community hospitals (Michigan Hospital Medicine Safety Consortium). Signs and symptoms were obtained from medical record review 3 days before and after urine-culture collection. Bacteremic UTI was defined as any positive blood culture growing at least 1 organism matching the urine culture. Risk factors for bacteremic UTI were assessed using multivariable logistic regression models with results expressed as odds ratios (ORs) for dichotomous variables and relative risks (RRs) for continuous variables. Results: Of 11,793 patients meeting study criteria, 73.6% were female with a median age of 78.2 years. Overall, 41.8% had AMS, 33.8% had dementia, 15.6% had an indwelling urinary catheter, and 54.6% had complicated urologic history (eg, urologic surgery). Of these, 166 patients (1.4%) developed bacteremic UTI. On adjusted analysis, male sex, hypotension, heart rate >90, urinary retention, fatigue, log of serum leukocytosis [1 log increase in serum WBC = 2.718 × serum white blood cell count (WBC)], and pyuria with >25 WBC per high-powered field (WBC/hpf) on urinalysis were associated with bacteremic UTI (Table). Older age, presence of an indwelling catheter, complicated urologic history, functional decline, AMS, dementia, and change in urine were not associated with higher odds for bacteremic UTI (Table). Of patients with AMS and no definitive signs or symptoms of a UTI, only 89 (1.8%) of 4,932 developed a bacteremic UTI. Conclusions: Bacteremic UTI is relatively rare in hospitalized inpatients presenting with bacteriuria without symptoms of UTI. Predictors of bacteremic UTI included male sex, hypotension, tachycardia, urinary retention, fatigue, serum leukocytosis, and higher levels of pyuria (>25 WBC/hpf) on urinalysis. Our findings provide stewards a framework to risk stratify inpatients of older age who present with positive urine cultures but without (or are unable to express) signs or symptoms of UTI.
Disclosures: None
Risk Factors and outcomes associated with inappropriate empiric broad-spectrum antibiotic use in hospitalized patients with community-acquired pneumonia
- Tejal Gandhi, Lindsay Petty, Valerie Vaughn, Anurag Malani, David Ratz, Tawny Czilok, Jennifer Horowitz, Elizabeth McLaughlin, Lisa Dumkow, Stephanie Burdick, Danielle Osterholzer, Mariam Younas, Steven Bernstein, Scott Flanders
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s31-s32
-
- Article
-
- You have access Access
- Open access
- Export citation
-
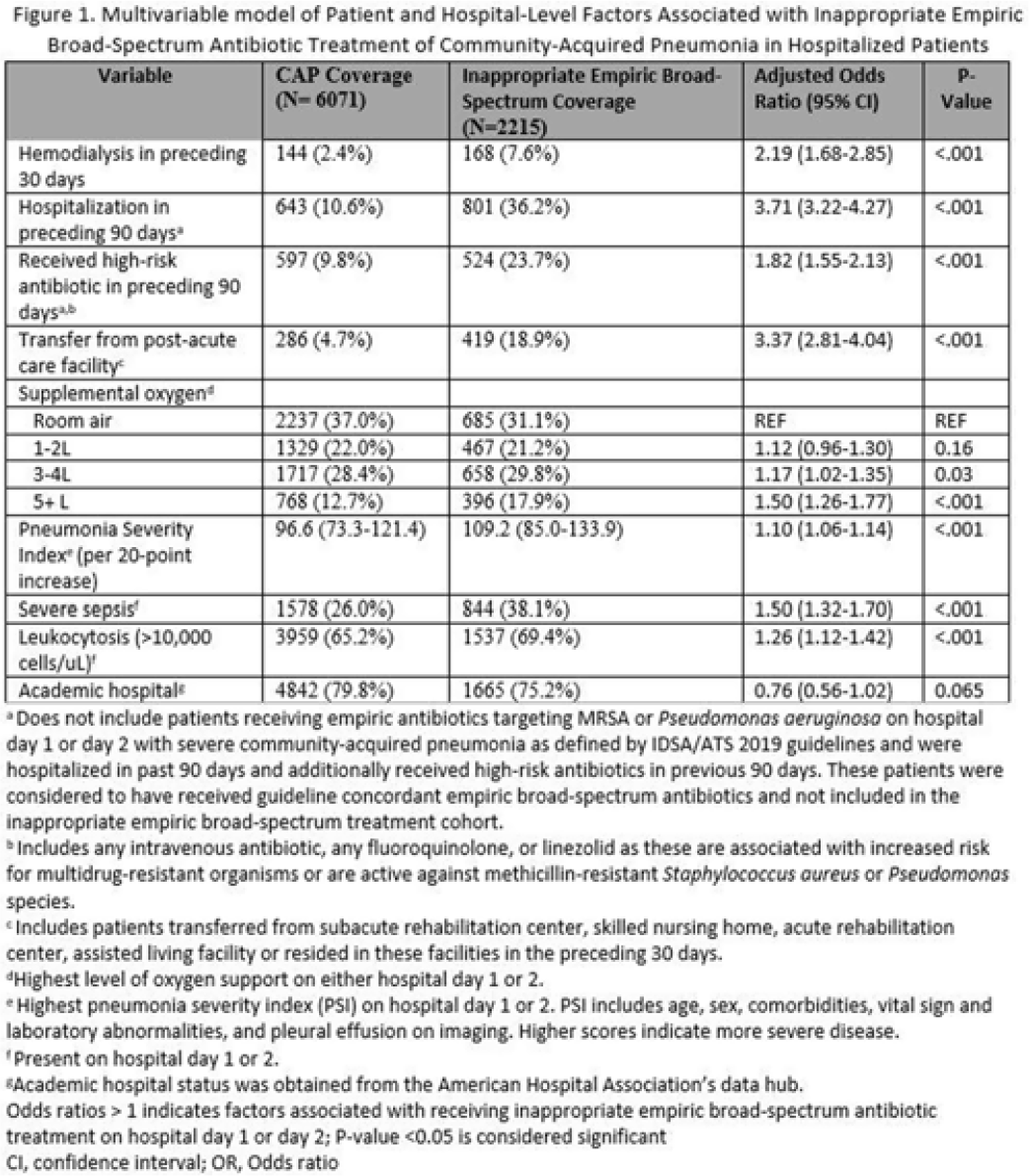
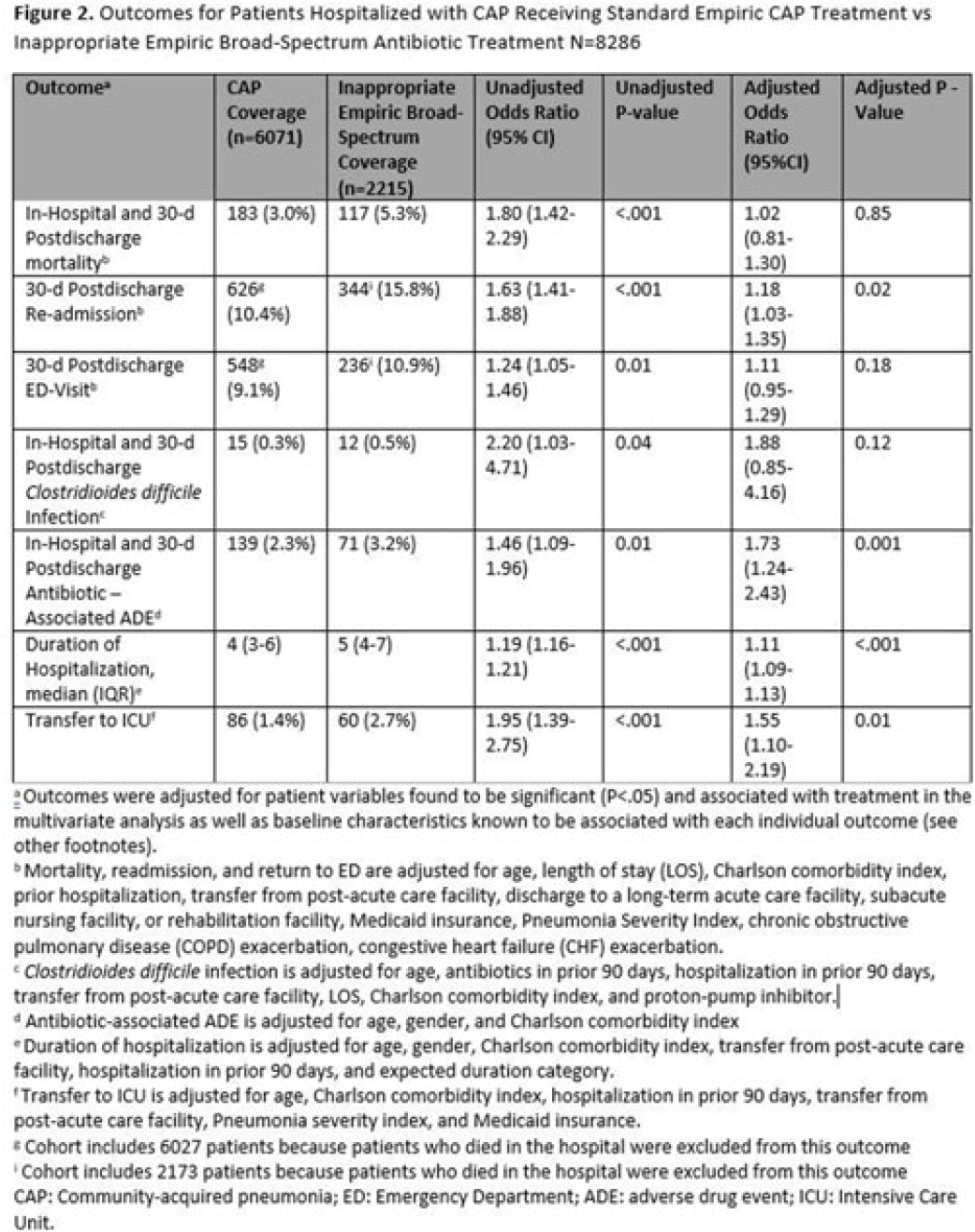
Background: Inappropriate broad-spectrum antibiotic use targeting methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa can result in increased adverse events, antibiotic resistance, and Clostridioides difficile infection. In 2019, revised ATS/IDSA community-acquired pneumonia (CAP) guidelines removed healthcare-associated pneumonia (HCAP) as a clinical entity and modified patient factors warranting empiric broad-spectrum antibiotic (BSA) use. As a result, most patients hospitalized with CAP should receive empiric antibiotics targeting standard CAP pathogens. Based on revised guidelines, we evaluated predictors and outcomes associated with inappropriate BSA use among hospitalized patients with CAP. Methods: Between November 2019 and July 2022, trained abstractors collected data on non-ICU adult medical patients admitted with CAP at 67 Michigan hospitals who received either an inappropriate empiric BSA on hospital day 1 or 2 or a standard CAP regimen. Inappropriate empiric BSA use was defined as use of an anti-MRSA or anti-pseudomonal antibiotic in a patient eligible for standard CAP coverage per IDSA guidelines. Patients with immune compromise, moderate or severe chronic obstructive pulmonary disease (COPD), pulmonary complication, or guideline-concordant treatment with BSA were excluded. Data collected included comorbidities, antibiotic use and hospitalizations in the preceding 90 days, cultures in the preceding year, signs or symptoms of pneumonia, hospital characteristics, and 30-day postdischarge patient outcomes. Data were collected through chart review and patient phone calls. Predictors of inappropriate empiric BSA were evaluated using logistic general estimating equation (GEE) models, accounting for hospital-level clustering. We assessed the effect of inappropriate empiric BSA (vs standard CAP therapy) on 30-day patient outcomes using logistic GEE models controlling for predictors associated with the outcome and probability of treatment. Results: Of 8,286 included patients with CAP, 2,215 (26.7%) were empirically treated with inappropriate BSA. The median BSA treatment was 3 days (IQR, 2.5). After adjustments, factors associated with inappropriate empiric BSA treatment included hospitalization or treatment with high-risk antibiotics in preceding 90 days, transfer from a postacute care facility, hemodialysis, support with ≥3 L supplemental oxygen, severe sepsis, leukocytosis, and higher pneumonia severity index (Fig. 1). After adjustments, patients with inappropriate empiric BSA treatment had higher readmissions 30 days after discharge, more transfers to the intensive care unit, more antibiotic-associated adverse events, and longer hospitalizations (Fig. 2). Conclusions: Patients hospitalized with CAP often received inappropriate BSA as empiric coverage, and this inappropriate antibiotic selection was associated with worse patient outcomes. To improve patient outcomes, stewardship efforts should focus on reducing inappropriate BSA use in patients hospitalized for CAP with historic HCAP risk factors or severe CAP without other guideline-directed indications for BSA.
Financial support. H.M.S. initiative is underwritten by Blue Cross and Blue Shield of Michigan.
Disclosures: None
Risk factors and outcomes associated with community-onset and hospital-acquired coinfection in patients hospitalized for coronavirus disease 2019 (COVID-19): A multihospital cohort study
- Part of
- Lindsay A. Petty, Scott A. Flanders, Valerie M. Vaughn, David Ratz, Megan O’Malley, Anurag N. Malani, Laraine Washer, Tae Kim, Keith E. Kocher, Scott Kaatz, Tawny Czilok, Elizabeth McLaughlin, Hallie C. Prescott, Vineet Chopra, Tejal Gandhi
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 9 / September 2022
- Published online by Cambridge University Press:
- 26 July 2021, pp. 1184-1193
- Print publication:
- September 2022
-
- Article
- Export citation
-
Background:
We sought to determine the incidence of community-onset and hospital-acquired coinfection in patients hospitalized with coronavirus disease 2019 (COVID-19) and to evaluate associated predictors and outcomes.
Methods:In this multicenter retrospective cohort study of patients hospitalized for COVID-19 from March 2020 to August 2020 across 38 Michigan hospitals, we assessed prevalence, predictors, and outcomes of community-onset and hospital-acquired coinfections. In-hospital and 60-day mortality, readmission, discharge to long-term care facility (LTCF), and mechanical ventilation duration were assessed for patients with versus without coinfection.
Results:Of 2,205 patients with COVID-19, 141 (6.4%) had a coinfection: 3.0% community onset and 3.4% hospital acquired. Of patients without coinfection, 64.9% received antibiotics. Community-onset coinfection predictors included admission from an LTCF (OR, 3.98; 95% CI, 2.34–6.76; P < .001) and admission to intensive care (OR, 4.34; 95% CI, 2.87–6.55; P < .001). Hospital-acquired coinfection predictors included fever (OR, 2.46; 95% CI, 1.15–5.27; P = .02) and advanced respiratory support (OR, 40.72; 95% CI, 13.49–122.93; P < .001). Patients with (vs without) community-onset coinfection had longer mechanical ventilation (OR, 3.31; 95% CI, 1.67–6.56; P = .001) and higher in-hospital mortality (OR, 1.90; 95% CI, 1.06–3.40; P = .03) and 60-day mortality (OR, 1.86; 95% CI, 1.05–3.29; P = .03). Patients with (vs without) hospital-acquired coinfection had higher discharge to LTCF (OR, 8.48; 95% CI, 3.30–21.76; P < .001), in-hospital mortality (OR, 4.17; 95% CI, 2.37–7.33; P ≤ .001), and 60-day mortality (OR, 3.66; 95% CI, 2.11–6.33; P ≤ .001).
Conclusion:Despite community-onset and hospital-acquired coinfection being uncommon, most patients hospitalized with COVID-19 received antibiotics. Admission from LTCF and to ICU were associated with increased risk of community-onset coinfection. Future studies should prospectively validate predictors of COVID-19 coinfection to facilitate the reduction of antibiotic use.







